Date Due: Wednesday, 30 June 2004
The total maximum points were 60. Point distribution for each question noted below.
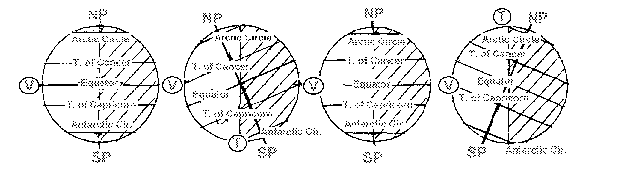
Assume that the sun is to the left of this page and produces a circle of illumination (the vertical lines) upon the globe for each of the four dates. Upon each globe:
a. Mark and label the North and South Poles.
b. Draw and label the Equator.
c. Draw and label the i.) Tropics of Cancer and Capricorn and ii.) the Arctic and Antarctic Circles.
d. Mark with the letter "V" that latitude where the sun appears to be directly overhead at local solar noon.
e. Mark with the letter "T" that latitude at the edge of the polar night where the sun appears to be just on the local horizon at local solar noon. (Hint: Where is the edge of polar night?)
(20 pts)
21 MARCH 21 JUNE 23 SEPTEMBER 23 DECEMBER
See also visible geosynchronous satellite images (courtesy of DataStreme and Project Atmosphere, the education initiative of the American Meteorological Society) that illustrate the effect:
21 MARCH (See 23 Sept.) 21 JUNE 23 SEPTEMBER 23 DECEMBER
a. The solar constant for the earth is approximately:
(2 pts)
|
1.97 cal per sq. cm. per min or 1372 Watts
per sq. meter. |
b. A mythical planet has an orbit with an average planet-sun distance exactly half that of the earth's. What would be the solar constant for this mythical planet? [Hint: make use of your answer from above.]
(4 pts)
|
The inverse square law means that the planet at one half the distance would have 4 times (or 2 squared) the amount of energy per unit area per unit time. 7.88 cal per sq. cm. per min or 5488 Watts per sq. meter. |
c. What is the planetary albedo of the planet earth?
(2 pts)
|
From lecture: |
Object A and Object B are ideal radiators. If A were hotter than B, then:
a. Which object would radiate more energy?
b. Which object would radiate more of its energy at a shorter wavelength?
(1 pt. each or 2 pts)
|
a. A (A consequence of Stefan-Boltzmann law) b. A (A consequence of Wien's Displacement law) |
Using the New Wind-Chill Chart in your textbook (page 92):
a. What is the wind chill equivalent temperature if the ambient air temperature were 15˚F and the wind speed were 10 mph?
b. What is the wind chill equivalent temperature if the ambient air temperature remained at 15˚F, but the wind speed increased to 20 mph?
c. What has caused the difference between your answers a and b above? Why?
d. To what temperature does your automobile reach in part a? in part b?
(12 pts)
|
From the Table: a. +3˚F is the wind chill equivalent temperature b. -4˚F is the wind chill equivalent temperature c. The increased wind speed causes the difference in the wind chill equivalent temperatures. The convective heat loss from the human body increases with increased winds. A statement about the air being colder is not correct since the ambient air temperature remains the same. d. The temperature of your automobile can only reach the ambient air temperature of 15˚F in both cases, and go no lower. The wind-chill equivalent temperature is not relevant here. However, in the second case, the stronger winds would hasten the cooling process, causing the engine block to cool to the ambient at a faster rate. |
-- [Please use the appropriate units!]
a. How much energy is required to entirely melt 1 gram of ice at the ice point?
b. How much energy is required to evaporate 1 gram of liquid water at room temperature?
c. How would the temperature of 1 kilogram of liquid water originally at 20˚C change if 5000 calories were used in the heating process (assume no phase transformations)? If a temperature change would take place, indicate the amount of change (and the direction of the temperature change).
[Please show your work and include units!]
(8 pts)
|
a. 80 cal (This value is called the latent heat of melting.) b. 590 cal (Note: The latent heat of evaporation is a function of temperature, being 590 cal at 20˚C and decreasing to 540 cal at 100˚C. The reason for this change in latent heat is that at room temperature water molecules are more tightly attracted to one another and would require more energy to break these "hydrogen bonds" of attraction than at higher temperatures when the water molecules are more active.) c. 5 Celsius degree heating Since 1 calorie is the heat needed to raise 1 gram of water 1 Celsius degree, then 5000 calories would raise 1000 grams by 5 Celsius degrees (making the liquid warm from 20˚C to 25˚C). |
7. Current Weather on the Web (8 pts)
See http://www.aos.wisc.edu/~hopkins/aos100/homework/s04hmk02k.html
This portion of the homework was designed to have you access to a
location on the Internet where you can find the times of sunrise and
sunset for not only Madison, but many locations throughout the country.
| Any
"reasonable answer" that fell within the range of values
for the week's sunset times in Madison was accepted. NOTE: Just by being outside in the early evening, you should have recognized that sunset has been about 8:40 PM CDT during the 2 weeks following the summer solstice. This relatively late sunset is enhanced by the observance of Daylight Saving Time. |
| The
earliest sunset of the year occurs at 1622 CST (or 4:22 PM)
during a 3-day span centered on 7-9 December 2004. The latest sunset of the year occurs at 1941 CST (or 7:41 PM) during a 11-day span centered on 21 June- 1 July 2004. Since Wisconsin, along with much of the rest of the nation observes Daylight Saving Time (from the first Sunday in April until the last Sunday in October), the time of the latest sunset would be 2041 CDT (or 8:41 PM). NOTE: The reason why the earliest sunset does not occur exactly on the winter solstice (about 21 December) and the exact date of the latest sunset does not coincide with the summer solstice (about 21 June), is that the sun is a poor time-keeper. The movement of the earth around the sun in an elliptical orbit along with the earth's spin axis being tilted from its orbital plane means that the apparent sun is several minutes ahead of clock time in December and minutes behind clock time in early July. |
Latest revision: 30 June 2004 (2300 UTC)
Produced by Edward J. Hopkins, Ph.D. Department of Atmospheric and Oceanic Sciences University of Wisconsin-Madison, Madison, WI 53706 hopkins@meteor.wisc.edu
URL Address: aos100/homework/s04hmk02a.html